pHasing into fatigue
An overview of exercise and acidity
“That is what you call trying to run through a sea of lactic acid” - commentator at the UK Athletics Championships as an athlete fell at the finish line of the 800m
Going “lactic” is common parlance for athletes and coaches. It’s that feeling of, well, pain and genuine discomfort. In an effort to run a fast 400, 800, and 1 mile this summer I have been frequently encountering this pain in training. Last weekend I dove head first in the sea of lactic acid during this workout:
4 sets of: 600m (1min rest) - 400m (1.5min rest) - 200m (5min rest)
Fatigue comes in many forms. The reason you want to stop during a maximal 800m run is a much different feeling than what makes you want to stop a marathon. We’re focusing on the short-duration pain.
Fatigue: when you lose the ability to produce as much force as you had been.
What’s causing it?
There are several contributing factors, but metabolic acidosis is certainly in the lineup. Before we get into it, a quick note on lactate and lactic acid.
Lactate and Lactic Acid
Lactic acid is the historical villain for causing metabolic acidosis and fatigue. However, it may not even exist in the body in relevant amounts (lactate and lactic acid are different molecules).
“To this day there remains a strong belief amongst exercise and sport physiologists, athletes and coaches, that lactic acidosis is the major villain underpinning fatigue. Despite this, a fundamental scientific point is that virtually no lactic acid appears in the body during exercise” - Simeon Cairns & Michael Lindinger
Article: Lactic acidosis: implications for human exercise performance
The debate is (or was) even more heated if you ask whether lactate/lactic acid causes fatigue. Lactate and acidosis tend to increase at the same time during fatiguing exercise, but correlation is not causality. You could even easily make the opposite interpretation, that lactate is increasing in an effort to prevent fatigue, as George Brooks would say.

Most people agree now that lactate does not cause fatigue. It is a remarkable carbohydrate molecule used for many, many things in the body. Without even getting into complex biochemistry, there are 2 very simple observations illustrating that lactate itself does not cause fatigue:
Lactate keeps increasing or stays elevated several minutes after exercise while your ability to produce force has recovered. Say you run maximally for 400 meters. Lactate will get pretty high. After 60 seconds you go again as hard as you can. Lactate is still high or even maximal, but I guarantee you’ll run the second 400 faster than if you hadn’t taken any rest. So there’s a disconnect between the ability to produce force and lactate levels.
When you take sodium-bicarbonate, high-intensity performance is improved while lactate goes higher than it would normally.
From previous blogs you’ll know that during high-intensity exercise we use mainly carbs for fuel. During this process each sugar molecule goes through a series of steps that produces energy along the way, and ultimately ends up with something called pyruvate. At this point, pyruvate is at fork in the road.
If the exercise is somewhat sustainable, it takes a longer, more scenic route that produces a bunch more energy. If intensity is really high (a maximal 800) and we’re demanding energy very fast (more than we can replenish), there’s no time for the scenic option and it takes a shortcut that gives us quick energy. That second option is getting converted into lactate, but it’s not sustainable.
One of the main benefits of the pyruvate-to-lactate conversion is producing NAD+, which keeps the sugar-breakdown process in full swing. Lactate can subsequently be used to produce energy itself in other muscle fibers, be sent to the liver to produce glucose, and other complex biochemical things.
But, to be clear, we’re always making lactate. You have a small amount in your blood right now. Most of the time we make use of it directly for energy and also converting it back into glucose. During intense exercise it starts to accumulate, and we produce more than we can dispose of. It’s really just a byproduct of carbohydrate breakdown. High intensity = high carb breakdown = high lactate. So we have this connection between exercise intensity and lactate, but where’s the acid?
Exercise Acidosis
Acidity is measured with the 0-14 pH scale. At rest, human blood pH is about 7.4 and can decrease as low as 6.9 during maximal rowing. In running and cycling the values can get down to about 7. Within human muscle cells, resting pH is about 7.1, with values as low as 6.3 (rowing) to 6.6 (running and cycling) after maximal exercise.
The further we go above a sustainable exercise intensity, the more acidic we get, and the more muscle function is impaired. With that being said, other things besides low pH contribute to fatigue, and from the previous sentence it’s hard to say acidosis explicitly causes fatigue (remember the lactate correlation is not causation example). But let’s look at how the acidity comes about and what its effects are.
One reason rowing (and carrying heavy groceries) feels so challenging is because the pH of our arms gets lower than the legs. Part of that is because the upper body has more fast-twitch muscle which is more prone to acidity. Rowers and 800m runners have more fast-twitch muscle than marathoners and can induce more acidity, but they also have ~50% or more greater capacity to buffer the acidity as a result of their specific training adaptations.
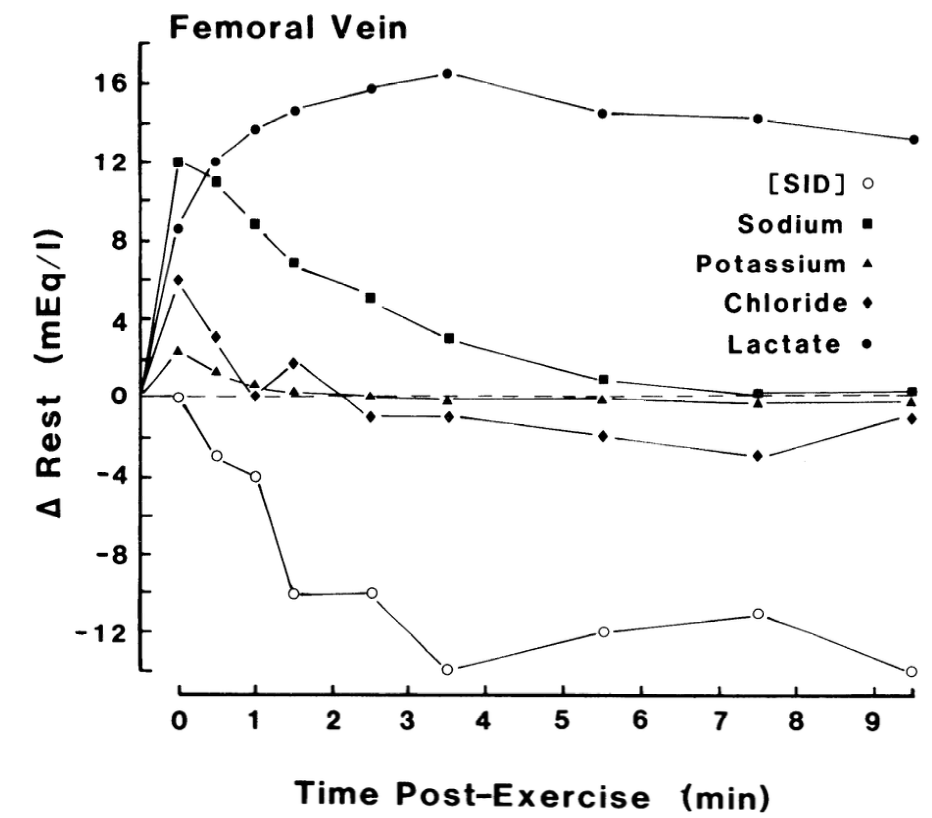
Continuing with our 800 meter run example, within seconds of the gun going off both anaerobic and aerobic metabolism kick into high gear. The metabolic reactions where we breakdown ATP (the energy currency of the body), phosphocreatine (PCr), and glycogen produce energy and cause changes in ions with positive and negative charges (stay with me now).
When you add up all of the positively and negatively charged molecules, you get the strong ion difference (SID). Lactate is negatively charged and goes up A LOT, but sodium for example is positively charged and also goes up. Potassium is positively charged but goes down. During intense exercise if you add all of these up the SID becomes negative (white circles in the figure above).
But, the body likes to remain neutral with these electrical charges floating around. To counteract all this negativity, hydrogen (H+, positive) increases to balance things out through the same metabolic reactions. pH stands for potential hydrogen, and increasing H+ decreases pH and vice versa.
How might this acidosis influence fatigue/performance?
One way is by inhibiting carbohydrate metabolism. When you give people an oral dose of ammonium chloride, which causes acidosis, then have them exercise, the activity of enzymes involved in breaking down carbs decreases and they use less glycogen. In that same study subjects also ventilated much more at the same intensity which (in my experiences) contributes to higher perceived effort.
Another is by directly interfering with the process of muscle contraction. On a cellular level the hydrogen gets inside the muscle machinery and interferes with calcium, which has important role in how we produce force.
In the picture below, scientists took out a piece of quadriceps muscle and measured how much power the individual muscle fibers could produce. They also looked at how fast the fibers could contract. They did this in the presence of increasing amounts of acidity. In each group, from left to right is the maximal power or velocity at muscle pH levels of 7.0, 6.8, 6.6, and 6.2. As acidity goes up, muscle power and velocity goes down. The top row is fast twitch fibers and the bottom row is slow twitch fibers.
If you looked at the figure closely you’d see that pH wasn't the only thing they manipulated. Inorganic phosphate (Pi), another byproduct of muscle contraction was also increased as the pH went down because that’s what happens in actual exercise.
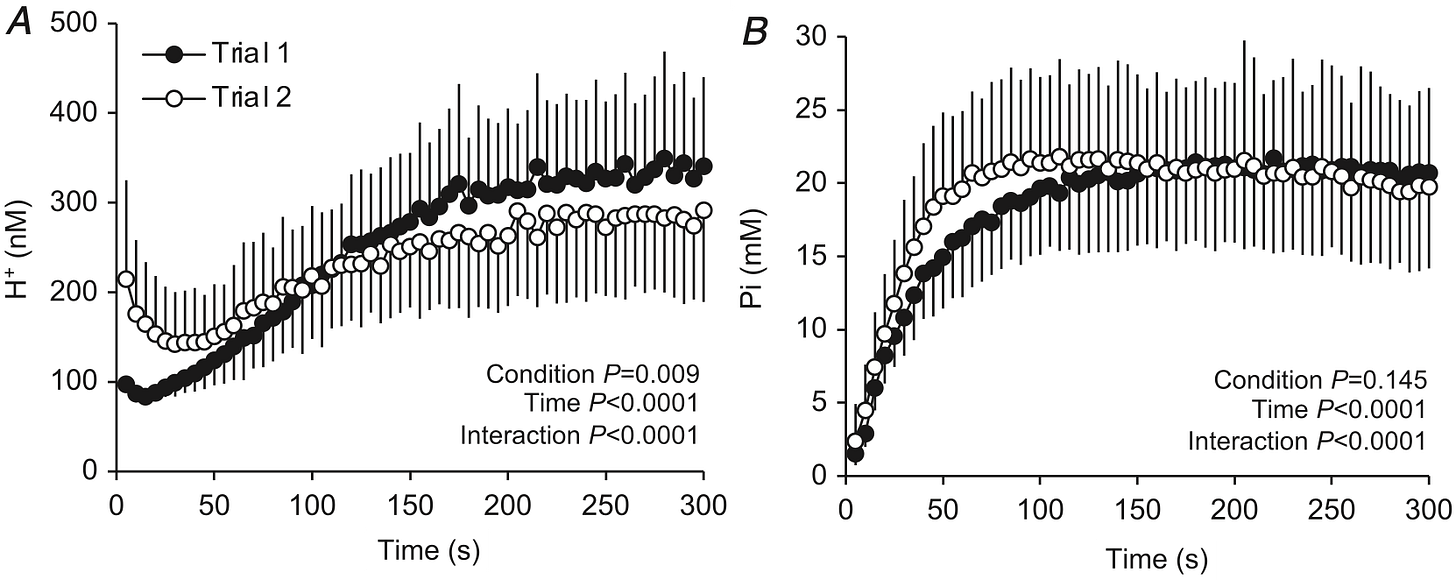
A study in humans exercising tried to see which one (pH or Pi) was more important by having people do 60 maximal muscle contractions over 5 minutes, rest for 5 minutes, then do it again. It was designed this way because pH would still be decreased (H+ still elevated) at the start of the second bout, but Pi returns to normal quickly.
In the study they were able to measure how much force the muscle itself had the capacity to produce by delivering an electrical stimulation to activate the muscle. But since the subjects were also maximally contracting their muscles, they could determine the voluntary muscle activation, which tells us how much effort they were able to muster from the brain to the muscle.
Basically, both of them matter and were directly related to reduced force production during both exercise bouts. But acidity specifically reduced voluntary muscle activation, indicating “central fatigue”. So the low pH is also sending a signal to the brain that makes us not want to contract our muscles as hard. Makes sense. It hurts!
To summarize, acidity 1) impairs carbohydrate metabolism, 2) interferes with muscle contraction, and 3) tells our brain to ease off.
Are there any benefits to acidity?
Of course! What fun would it be if everything was so cut and dry. When our blood gets acidic, it helps us dump oxygen off of hemoglobin in red blood cells. This is good for exercise when we are trying to get oxygen from the air, into the blood, and ultimately into our muscles for energy.
About 20 years ago a Petri dish study on rat muscle fibers suggested that acidity helped resist fatigue, showing that acidic conditions helped the muscle remain more excitable and willing/ready to contract. It had to do with counteracting the negative effects of potassium accumulation on muscle fatigue. I don’t think their data was wrong, it’s just the applicability to exercising humans which is hard to tease out.
Clearly, the science of fatigue can be complicated, but at the end of the day we have great tools to manage it regardless of the cause, primarily hard training specific to the demands of an event. Stay tuned for my next article where I’ll go into supplement strategies that might provide an extra edge.
Some sources
Buffering capacity of deproteinized human vastus lateralis muscle. J Appl Physiol. 1985. (this study compares muscle fiber type and buffer capacity in rowers, 800m runners, and marathon runners).
Effect of induced metabolic acidosis on human skeletal muscle metabolism during exercise. Am J Physiol. 1999. (this study induced acidosis with ammonium chloride and looked at carbohydrate metabolism)
Cumulative effects of H+ and Pi on force and power of skeletal muscle fibres from young and older adults. J Physiol. 2025 (this study looked at power and velocity on muscle fibers during acidic conditions)
On the role of skeletal muscle acidosis and inorganic phosphates as determinants of central and peripheral fatigue: A 31 P-MRS study. J Physiol. 2022. (this is the Pi versus acidity study)
Lactic acid--the latest performance-enhancing drug. Science. 2004 (this short article covers the more complex article suggesting acidity prevents fatigue)
Sean Langan has his PhD in exercise physiology. He is interested in how exercise training rewires our biology and harnessing the intersection of human physiology, technology, research, and coaching to improve human performance.




Great read!!